Volume 2 - Year 2014 - Pages 96-102
DOI: 10.11159/ijepr.2014.014
A Feasibility Study of a Clay-Zeolite Media as a Removal Technology of Total Phosphorus from Wastewater
Amanda L. Ciosek and Grace K. Luk
Ryerson University, 350 Victoria Street, Toronto, Ontario, M5B 2K3, Canada
amanda.alaica@ryerson.ca, gluk@ryerson.ca
Abstract - Phosphorus is a major factor in the eutrophication process, modifying water and habitat at high concentrations. This paper describes the development of a cost-effective, efficient, and affordable on-site Total Phosphorus (TP) removal technology. By investigating the chemical adsorption of a clay-zeolite media, the objective was to demonstrate TP removal capacity with a primary focus on pellet media design to overall removal efficiency. The investigation included pellet formation, size, conditioning, furnace exposure temperature and duration, as well as initial influent concentration (theoretical septic tank effluent range of 6-20 mg/L), with a response-contact time of 45 minutes. The research followed various stages of optimization and consisted of a scaled-down testing unit based on a 1.91 cm pellet diameter construction, for a 45 minute detention time, and achieved a removal of approximately 45%.
Keywords: Total Phosphorus, Eutrophication, Adsorption, Zeolite.
© Copyright 2014 Authors - This is an Open Access article published under the Creative Commons Attribution License terms. Unrestricted use, distribution, and reproduction in any medium are permitted, provided the original work is properly cited.
Date Received: 2013-09-18
Date Accepted: 2014-09-04
Date Published: 2014-10-03
1. Introduction
A world water crisis is growing as clean drinking water availability diminishes. Toxic effects on ecosystems from untreated sewage discharge and land runoff have contributed to another growing global issue - eutrophication. Eutrophication is how the ecosystem retorts to activities that infect water bodies with phosphorus and other nutrients; modifying animal and plan populations, as well as their water and habitat quality [3].
Sensitive areas prone to phosphorus contamination from domestic sewage must meet stringent effluent limits, following surface and subsurface disposal, in order to control the eutrophication process problem. The Ontario Ministry of Environment standards, outlined in The Lake Simcoe - Phosphorus Reduction Strategy, must be employed in order to meet the current baseline compliance limit of 0.30 mgP/L. The current research focuses on the on-site aerobic treatment of domestic wastewater, and the analysis of the overall removal efficiency was conducted by employing the Langmuir and Freundlich Adsorption isotherm models.
There are various removal technologies, involving biological, chemical, and physical processes. In order to make an effective engineering decision, a stringent screening methodology was considered [1]. The physio-chemical removal process was selected based on its consistency, and industry acceptability.
The adsorption process where molecules from a gas or solution phase bind to a condensed layer on a solid or liquid surface was the basis for experimentation. Adsorption isotherms are a graphical representation of the quantity adsorbed as a function of pressure of the gas or solution [5]. The most significant model of mono-layer adsorption is given by the Langmuir Adsorption Isotherm, whereas the Freundlich Adsorption Isotherm [9] is a modification of it to involve a multisite analysis [5]. Ion-exchange is a function of the degree of substitution of aluminium for silicon in the framework structure [6]. A major attribute of zeolites is their ability to exchange ions with external media [10]. The process of adsorption was explored using a clay-zeolite media, which possesses natural adsorptive and ionic chargeable properties.
A source-specific action was taken by developing an on-site phosphorus removal unit. The purpose was to obtain a cost-effective, efficient, and affordable technology, accessible to individual facility owners. This advancement is unique to the industry, in that it offers a small footprint, with naturally available raw materials, and a modular design. This development created a means of reducing the phosphorus of natural water bodies, thereby overcoming issues associated with eutrophication.
2. Materials and Methods
A small testing unit was constructed in order to assess the performance of the various pellet types. The removal efficiency relative to initial concentration and contact time was the basis for analysis and optimization. The testing unit is made of two identical systems of dual cylindrical compartments, constructed with schedule 40 PVC tubing of 7.62 cm diameter and 15.24 cm height.
Bentonite clay, kaoline clay, zeolite (sieve mesh distinctions 8x40 and 14x40) gradations, and deionized distilled water of various ratios were investigated. A preliminary method involved incorporating primarily moist clay and zeolite particulates, to form a 1.91 cm diameter spherical pellet with a melon baller. These pellets were air dried for 24 hours, followed by furnace exposure at 100oC and dehydrated for 24 hours, then gradually increased to 600oC, allowing for calcination for 24 hours. Furthermore, another pellet composition was developed from the work of Jasra et al. [7]. Due to material limitations and porosity objectivity (coarser zeolite), the amount of clay was modified based on feasibility; overall clay incorporated into the mix was 80% of zeolite, but still analyzing as described in [7] with a two clay combination of bentonite and kaoline (hydrous aluminum silicate). A total of 7 pellet types were formed; six with the bentonite-kaoline blend and varied zeolite gradations, and one with the moist clay of coarser gradation.
3. Experimental Development
A protocol was developed in order to determine the optimal pellet type. The optimization testing unit was employed by analyzing a 1.91 cm pellet diameter.
According to Song [8], two random packing factors occur; close (RCP) and loose (RLP). Their respective maximum density, termed the Atomic Packing Factors (APF), are 64% (RCP) and 55% (RLP), respectively. The medium of these values was selected at 60%, as stated in Eq. (1) [2]:
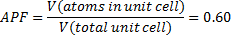
With the assumption of a 60% atomic packing factor, the volume of the pellets (VP) to occupy the volume of the testing unit (Vtu) would be expressed in Eq. (2), as follows:
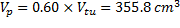
In order to determine the most feasible pellet size, various extraction and pelletizing methods were explored, including a pelletizing machine, truffle moulds, and measuring spoons. From the preliminary testing of the consistency of the original procedure, 6 pellets formed by hand were randomly selected to measure their diameters, with an average of 1.83 cm.
Various measuring spoons shown in Fig. 1 were analyzed, converting the volume of half a sphere, and translating it to its corresponding diameter of a complete sphere; the ½ teaspoon measurement corresponding to a 1.98 cm diameter. The average pellet and ½ teaspoon measuring spoon diameters are 1.83 cm and 1.98 cm, respectively. These values bound a 1.91 cm pellet diameter volume. For repeatability purposes, a melon-baller of the corresponding diameter-volume was used for pellet formation. All calculations that followed correspond to a 1.91 cm pellet diameter volume.

To determine the number of pellets to fill the testing unit, the corrected pellet volume was employed of a 1.91 cm diameter pellet in Eq. (3) asfollows:
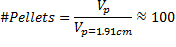
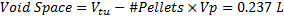
This void space corresponds to the sump volume for experimentation; a 25% safety factor was applied, such that 0.296 L was carried through testing. In order to account for time and economic factors to the experiment, specific initial influent concentrations and contact times (converted to flow rate) were selected. A septic tank's effluent (apparatus influent) concentration of phosphorus ranges approximately from 6 to 20 mgP/L [4].
The following subsections outline the actions taken and the decisions made over the course of each of the three design stages.
3.1. Pre-Optimization Stage
The lower (6 mgP/L), middle (14 mgP/L) and upper (18 mgP/L) bounds of initial influent concentrations were analyzed from the various pellet types. Given that only two contact times were explored (15 and 45 minutes) at this stage, the samples were extracted at these times rather than regenerating the testing unit with influent. The experimental design was modified due to the constraints of the TP meter (Orbeco MC500 Colorimeter Procedure - 326-Phosphate, Total). In addition, the pellets were conditioned at maximum solubility with calcium hydroxide brine to observe the effects on the removal efficiency.
3.2. Optimization Stage
Following the discoveries of Pre-Optimization, the furnace exposure became the major parameter of investigation. In addition, further exploration of conditioning versus as-received pellet form was introduced. This stage investigated the effects of conditioning concentration, as well as furnace exposure (in terms of both temperature and duration) against the minimum and maximum influent concentrations at two different contact times.
Approximately 300 pellets of both Type 6 (clay blend, fine zeolite) and Type 7 (moist clay, coarse zeolite) were formed; such that 100 non-conditioned, 100 conditioned with Calcium Hydroxide (Ca(OH)2) (C1) and 100 conditioned with Sodium Chloride (NaCl) (C2). The pellets were exposed to the furnace times of 6 hours at 100oC and 600oC (Phase I), and 12 hours at 100oC and 600oC (Phase II). The pellets conditioned were exposed to a low (A) and high (B) concentration or maximum solubility of both the Calcium Hydroxide (Lime) and Sodium Chloride (Salt) brine solutions. The pellets were submerged in a minimum and maximum influent, 6 mgP/L and 18 mgP/L, respectively. The effluent was drawn at two different contact times; 15 and 45 minutes.
4. Results
4.1. Pre-Optimization
The preliminary stage of the experiments began with testing the Type 7 pellet, at 6, 14, and 18 mgP/L influent concentrations. These concentrations were analyzed at 7.5 and 15 minute contact times. All readings indicated over-range for the TP meter; beyond the upper bound of its 0.02-1.1 mgP/L range. Consequently, both the 20 mgP/L stock (based on a 10,000 ppm phosphorus reagent) and 6 mgP/L influents were tested through various 50% dilutions, all of which were detectable.
This was followed by the analysis of the Type 6 pellet at the minimum influent concentration (6 mgP/L) and maximum contact time (15 and 30 minutes); once again, with a 90% removal assumed and no dilutions employed, all readings were undetectable by the TP meter. This was followed by the analysis of both the influent and effluent of Type 6 and 7 pellets, with minimum influent concentration (6 mgP/L), and maximum contact time (15 minutes). At this stage, the influent and effluent were exposed to 4- and 3-50% dilutions as in Table 1 and 2, respectively. Subsequently, these same pellets were conditioned with calcium hydroxide; there was a successful reading with dilutions. Table 3 provides a summary of the Pre-Optimization results, which is based on an influent concentration of 6 mgP/L for a contact time of 15 minutes.
Table 1. Influent dilution
| 4-50% Dilutions | 5-50% Dilutions | 6-50% Dilutions | ||
| Influent | ||||
| Conc. (mgP/L) | 6 | 12 | 18 | |
| Vol. (mL) | 0.95 | 0.50 | 0.25 | |
| Diluted Influent | ||||
| Conc. (mgP/L) | 0.375 | 0.375 | 0.281 | |
| Vol. (mL) | 15.2 | 16.0 | 16.0 | |
| DDW (mL)* | 14.25 | 15.50 | 15.75 | |
| 3-50% Dilutions | 4-50% Dilutions | 5-50% Dilutions | ||
| Effluent | ||||
| Conc. (mgP/L) | 6 | 12 | 18 | |
| Vol. (mL) | 1.9 | 0.95 | 0.50 | |
| Diluted Effluent | ||||
| Conc. (mgP/L) | 0.75 | 0.75 | 0.563 | |
| Vol. (mL) | 15.2 | 15.2 | 16.0 | |
| DDW (mL)* | 13.30 | 14.25 | 15.50 | |
| *DDW Volume = Diluted Volume - Influent Volume | ||||
Table 3. Pre-optimization pellet results summary
| Pellet Type | ||
| TP Removal | ||
| (mgP/L) | (%) | |
| 6 | ||
| Non-conditioned | 0.68 | 9.93 |
| Conditioned | 0.52 | 7.18 |
| 7 | ||
| Non-conditioned | 1.16 | 16.56 |
| Conditioned | 2.36 | 30.87 |
4.2. Optimization
Pre-Optimization confirmed that dilutions were required to receive TP Meter detection due to the reading restraints of the narrow range (0.02-1.1 mgP/L). Tables 1 and 2 were employed to the influent and effluent, respectively, of the optimization stage as well.
It is to be noted that when the pellets were exposed to the influent and minimum conditioning brines (C1A and C2A), a distinct sizzling sound occurred due to gas formation, and the pellets began to expand. In particular, when Pellet Type 7 was exposed to Phase I, the pellets began to 'explode' and fell apart. This is most likely due to the compact, dense attribute of the pellets. Consequently, Phase I analysis proceeded, utilizing all pellet fragments in the experiments. The surface area and contact points were greater, which may have skewed the results in its favour with greater phosphorus removal. This fragmentation would compromise the pellet integrity. Consequently, it was decided that Phase II furnace exposure be observed, thereby ensuring a more stable pellet composition.
Pellet types 6 and 7 were formed, for both Phase I and II furnace exposures, and carried through for experimentation. The final pellet type selected was the non-conditioned (NC) Type 7, Phase II (12 hour furnace exposure). This selection was based on the minor difference in overall phosphorus removal, taking into account the environmental and economic impacts of conditioning. This pellet type was recreated and tested once again, and its results demonstrated consistency; 40% removal was achieved with an 18 mgP/L influent concentration and 45 minute contact time.
5. Analysis
5.1. Pre-Optimization
Pellet Types 1 to 4 were not carried through to the quantitative analysis stage. These pellets were only formed for qualitative purposes; to observe the workability. Given that Types 1 to 4 were only of one clay type (Bentonite and Kaoline) rather than a blend (Types 5 and 6), the overall composition was brittle and consequently not considered ideal for mass production.
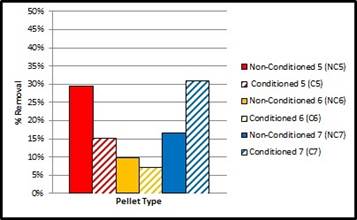
Pellet Types 5 and 6 were very porous compared to Type 7. Notably, Types 6 and 7 were exposed to more influent cycles than Type 5, during the preliminary stages of TP Meter detections; in order to refine the dilution calculations. As observed in Fig. 2, non-conditioned pellet Type 5 performed similar to that of conditioned pellet Type 7. Based on the coarser, zeolite gradation used to compose Type 5, the fluid dispersion was greater, such that the influent was able to traverse more through the material than the denser pellets Type 6 and 7. However, Type 5 removal was reduced once conditioned. For comparative purposes, the more compact pellets were carried further into analysis (Types 6 and 7), concluding that conditioned pellet Type 7 produced the highest removal efficiency from the pre-optimization perspective.
5.2. Optimization
5.2.1. Contact Time
Fig. 3 indicates that the removal for Pellet Type 6 is greatest with the Hydrated Lime Brine at maximum concentration (C1B), whereas Type 7 is more or less consistent whether non-conditioned or exposed to either conditioning solution at minimum concentration (C1A, C2A). The optimal removal of both pellet types is close in value.
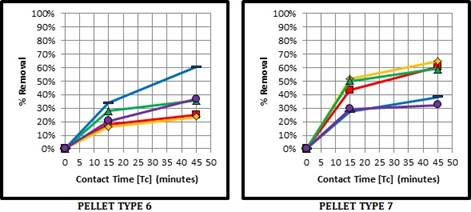

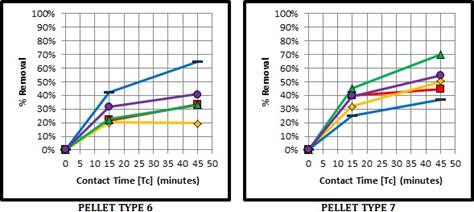

As shown in Fig. 4, Pellet Type 6 in phase II is optimal as in phase I; at a maximum Calcium Hydroxide solution concentration (C1B). For Type 7, the removal is greatest with once again the Salt Brine at minimum concentration, closely followed by its maximum concentration. Based on the physio-chemical process of adsorption, it is important to note that there are various exchangeable cations within the zeolite structure, including calcium and sodium. By applying the lime and salt conditioning methods, the goal was to enhance the cation exchange capacity (CEC) of zeolite to remove the phosphorus anions in solution. Figures 3 and 4 display the CEC performance of Pellet Type 6, and how this pre-treatment has improved its adsorption performance to reach that of Pellet Type 7 in its natural state.
5.2.2. Influent Concentration and Furnace Exposure
Pellet Type 7 overall removal is greatest among the two, taking into account the various contact times and furnace exposures. A key observation of the influent concentration results is that the removal was consistent, given the wide range of 6 to 18 mgP/L. This supports the conclusion that the septic tank effluent concentrations were addressed most effectively.
The Phase II furnace exposure removal is slightly greater among the various contact times and influent concentrations.
5.2.3. Optimization Analysis Summary
This subsection provides an overview of the major findings from the optimization stage. Table 4 outlines the percent removal at 45 minutes. For review, NC, C1A, C2A, C1B, and C2B refer to non-conditioned (as received), minimum (A) Lime (1) and Salt (2) Brines, and maximum (B) Lime (1) and Salt (2) Brines, respectively.
Table 4. Optimization 45 Minute Percent Removal Analysis
| Phase I | Phase II | Phase I | Phase II | ||||||
| Ci (mgP/L) | 6 | 18 | 6 | 18 | Ci (mgP/L) | 6 | 18 | 6 | 18 |
| Pellet Type 6 | Pellet Type 7 | ||||||||
| NC | 55 | 25 | 61 | 33 | NC | 74 | 60 | 44 | 45 |
| C1A | 18 | 24 | 24 | 19 | C1A | 38 | 65 | 29 | 50 |
| C2A | 55 | 36 | 52 | 33 | C2A | 59 | 59 | 70 | 70 |
| C1B | 39 | 60 | 36 | 65 | C1B | 25 | 38 | 24 | 37 |
| C2B | 44 | 37 | 43 | 41 | C2B | 44 | 32 | 55 | 55 |
With the contact time parameter analysis at the minimum and maximum influent concentrations and furnace exposures, it was determined that the performance of the non-conditioned (NC) or minimum Salt Brined (C2A) pellets were more superior, with pellet type 7 as optimal; presumably due to greater surface contact and overall ion exchange. The influent concentration parameter analysis confirmed also that Pellet Type 7 had a slight advantage in overall removal. The furnace exposure study revealed that Phase II removal is not only slightly greater, but also ensures a more stable product. As a result of the analyses, the final pellet composition was decided as Type 7 NCII taking into account economic and environmental impacts of conditioning, producing an optimization removal of 45% at the 45 minute contact time.
6. Conclusion
The overall objective of this research was to further the development of a cost-effective, efficient, and affordable on-site Total Phosphorus (TP) removal unit. Employing chemical adsorption technology, the clay-zeolite media was analyzed for a pilot unit to demonstrate the TP removal capacity from an artificial influent, simulating domestic wastewater effluent concentrations. The analysis to overall removal efficiency was based on media composition and formation, conditioning, influent concentration, and contact time exposure.
The analyses determined that Pellet Type 7 with Salt Brine conditioning solution at minimum concentration (C2A) and Phase II (12 hour furnace exposure) provided the greatest removal efficiency of 70%. However, the 25% removal efficiency difference among the non-conditioned and conditioned results was not substantial to account for the significant future impacts. As a result, from an economic and environmental perspective, the as-received (non-conditioned) Pellet Type 7 was deemed optimal for final mass fabrication.
There were key observations made during optimization. With regards to the influent concentration analysis, the septic tank effluent concentration range was applied to the pellets with a minimum and maximum concentration of 6 and 18 mgP/L, respectively. The investigation demonstrated that the extreme influent bounds provided similar removals; indicating an effective system.
There are various recommendations that can be drawn from this research. It should be noted that all pellets were analysed based on a single-feed, influent to pellet exposure. Further observations into multi-feed systems and how this would impact the pellets' performance would be useful for future investigations. Environmental conditions such as moisture and humidity, and freeze-thaw cycles should be investigated, which would better represent the field conditions. The field testing should also look into the life-cycle analysis of an apparatus, and to determine at what point would the media need to be either regenerated or replaced. With regards to regeneration, the conditioning brine disposal must be taken into consideration to prevent any adverse effects on the environment. In terms of replacement, the spent pellet material must be studied with the possibility for further use, such as fertilizer and its effects on the environment (i.e. leaching into the soil, and subsequently into the waterways). The research work presented an environmentally sustainable solution, and contributes to the development of a total phosphorus reduction strategy to restore, improve and protect our environment.
Acknowledgements
The authors would like to thank the Ontario Centres of Excellence and our industry partner, Virtual Engineers, for their financial and technical support of the project.
References
[1] Bowker, R. P. G., Stensel, H. D. (1990). Phosphorus Removal from Wastewater. Pollution Technology Review No. 189. Noyes Data Corporation. View Book
[2] Callister, W. D. (2007). Materials Science and Engineering-An Introduction. 7th Edition. Wiley & Sons, Inc. View Book
[3] Cloern, J. E., Krantz, T., Hogan, C. M., Duffy, J. M. (2010). Eutrophication. Encyclopedia of Earth. Eds. Cutler J. Cleveland. Environmental Information Coalition, National Council for Science and the Environment. Retrieved 03/05/11 from: View Article
[4] EPA. (2012). Onsite Wastewater Treatment Systems Technology Fact Sheet 8 - Enhanced Nutrient Removal - Phosphorus. EPA 625/R-00/008. Retrieved 01/04/2011 from http://www.epa.gov/nrmrl/pubs/625r00008/html/tfs8.htm
[5] Masel, R. I. (1996). Principles of Adsorption and Reaction on Solid Surfaces. John Wiley & Sons, Inc. pp. 235-248. View Book
[6] Mumpton, F.A. (1977). Mineralogy and Geology of Natural Zeolites. The College of Brockport. Mineralogical Society of America. Vol. 4. View Book
[7] Jasra, R. V., Tyagi, B., Badheka, Y. M., Choudary, V. N., Bhat, T. S. G. (2003). Effect of Clay Binder on Sorption and Catalytic Properties of Zeolite Pellets. Central Salt and Marine Chemical Research Institute. Indian Petrochemical Corp. Ltd. American Chemical Society. Indian Engineering Chemical Research. Vol. 42. pp. 3263-3272. View Article
[8] Song, C. et al. (2008). A phase diagram for jammed matter. Vol 453. Nature Publishing Group. View Article
[9] Sun, X., Jinzhu, H. (2010). The phosphorus-adsorption capacity and influencing factors of Zeolite. Advanced Materials Research Vols. 113-114. pp. 766-769. View Article
[10] Wang, S., Peng, Y. (2010). Natural zeolites as effective adsorbents in water and wastewater treatment. Chemical Engineering Journal Vol. 156. pp. 11-24. View Article

