Volume 2 - Year 2014 - Pages 80-88
DOI: 10.11159/ijepr.2014.012
The Environmental Impact of Urbanization within Abu Dhabi on the Microbial Profile of Man-Made Beaches
Fatima Al Marzooqi and Fatme Al Anouti
Zayed University, College of Sustainability Science and Humanities,
Department of Natural Science and Public Health
Khalifa City, Abu Dhabi, UAE
U2880028@zu.ac.ae, Fatme.AlAnouti@zu.ac.ae
Abstract - The United Arab Emirates (UAE) is witnessing a significant development in several areas. Many new construction projects are currently underway. One of the main project aims at establishing new beaches around the coastal areas of the UAE. Particularly, in Abu Dhabi the capital of UAE, a new man-made beach has been recently developed in the north coast of the city. The urbanization of this newly developed beach might have a significant impact on the environment. This study aims to investigate the effects of urban development of beaches within Abu Dhabi on the microbial profile of the soil. It is hypothesized that microbial growth would be higher in man-made beaches as compared to natural beaches. Methodology involved collection of 16 soil samples from two different beaches with emphasis on two elements: water content in soil and depth of soil. Subsequently, samples were processed and used for microbial cultivation using selective and differential growth media for the identification of some commonly encountered microorganisms namely "yeast, faecal coliforms, Escherichia coli and Staphylococcus aureus". Polymerase Chain Reaction (PCR) and electrophoresis were also used for further analysis. Results of the study revealed the existence of Staphylococcus aureus, Escherichia. coli and coliforms in the soil samples isolated from the man-made beach. Moreover, the hypothesis was supported by the results which showed higher microbial growth (CFU/g) for both factors: (depth of the soil and water content) for man-made beaches as compared to natural beaches.
Keywords: Microbiology, Beach, Abu Dhabi, Environment, Urbanization, Bacteria
© Copyright 2014 Authors - This is an Open Access article published under the Creative Commons Attribution License terms. Unrestricted use, distribution, and reproduction in any medium are permitted, provided the original work is properly cited.
Date Received: 2013-10-30
Date Accepted: 2014-04-11
Date Published: 2014-10-03
1. Introduction
Microorganisms have huge effects on ecosystem processes that sustain life on earth. They produce oxygen, store carbon and serve as essential factors in the nutrient cycle [14]. Species usually live together and depend on each other to survive. Rarely can we find ecosystems with little diversity. Each ecosystem has its own combination or mixture of microorganisms. For example, bacteria often occur at higher levels in the soil than in water. Also, they survive in the soil longer than they do in the water [29]. Nevertheless, few studies show that the bacteria survive longer in wet soil as compared to dry soil [30]. Many factors such as temperature, depth, moisture, air, organic matter availability, light availability, pH and size of soil particles play an important role in promoting the growth of microorganisms.
To maintain the normal cycle of life, the ecosystem should function normally. However, if the ecosystem loses one of it is vital components, detrimental effects could result. Many studies related to soil microbiology have attempted to explore the potential pathogenicity of microorganisms in beach soil. The World Health Organization (WHO) in 2003 [35] indicated that there was no epidemiological evidence to show the correlation between soils contaminated with microorganisms and human infection. Furthermore, Prado et al., [27] and Brandão et al., [5] in their studies demonstrated a lack of correlation between health and potentially pathogenic fungi inhabiting beach soil. However, Brandão et al., [5] mentioned that people sometimes are responsible for contaminating beach sands by carrying microorganisms that are left on the soil. Also humans could play a role by discarding organic waste that serves as food for the development of yeasts, bacteria, and molds. To some extent, those microorganisms will not cause severe effects to human unless they reach a certain level. Therefore, the level of contamination in beach soil should be known to ensure public safety.
To determine the level of contamination in beach soil, the faecal index of organisms is often used as a reliable indicator of the level of non-potentially pathogenic microorganism. These microorganisms are usually present in higher numbers as compared to the potentially pathogenic microorganisms and are easy to isolate, identify and count. Species included in the faecal index organisms are coliforms which consist of total coliforms, thermotolerant coliforms and E. coli; intestinal enterococci, clostridia and bacteriophages. One of the recent studies conducted in Portugal regarding microbial quality of beach soilrevealed the presence of potentially pathogenic fungi, yeasts, dermatophytes, total coliforms, intestinal enterococci and E. coli in soil samples taken from thirty three public beaches around the country. In this study, the investigators proposed a safe threshold for the potentially pathogenic microorganisms. They proposed 15 colony forming unit per gram (CFU/g) of soil for yeast, 17 CFU/g for potentially pathogenic fungi, 8 CFU/g for dermatophytes, 84 CFU/g for coliforms, 250 CFU/g for E. coli, and 100 CFU/g for intestinal enterococci. Potential health risks for human could occur if those thresholds are exceeded [30].
Bacterial species are important to determine the potential risk level of beach soil. Staphylococcus spp. is a flora which is widely spread in beach soil [8]. Examination of soil samples from two popular beaches in Chile showed that from a total of 85 strains of Gram-positive bacteria detected; cocci were the highest in number [27]. This was speculated to be the result of human activity in beaches as some individuals might carry this species on their skin. Another study in Portugal isolated P. aeruginosa and Clostridium perfringens from the soil of different beaches around the country. The investigators suggested that Clostridium perfringens could be a good faecal index microorganism [21].
With regards to potential pathogenicity of fungi in beach soil, most fungal diseases are associated with immune-compromised conditions [6, 21]. In the central coastal area of Portugal, dermatophytes showed a high percentage of around 42% in the beach soil. While others were T. rubrum, Trichophyton mentagrophytes and Microsporum nanum, all isolated from soil with low organic nutrient [33]. These fungi are all potentially pathogenic and could hence have serious implications on the health of beach users.
Another important factor to determine when studying microorganisms in beach soil is the water content in the soil. Brandão et al., [5] showed that the microbiological contamination is higher in dry soil than in wet soil. Several others also showed that contamination of soil was the highest in dry soil and during the summer than during spring [20, 31]. However, others also showed that microorganisms survive longer in wet soil [30]. Therefore, there is no clear conclusion whether growth of microorganisms is strictly higher in dry or wet soil. Concerning the correlation between species on wet and dry soil, it was found in Italy by Bonadonna et al., 2002 that a correlation exist between E. coli and enterococci; yeasts and molds; clostridial spores and staphylococci and between enterococci and spores of sulfite-reducing Clostridium [35]. While, Papadakis et al. [26] found no correlation between Staphylococcus aureus counts and the indices of faecal pollution counted from the wet soil of the beach [35].
All those species detected in the beach soil are associated with several factors such as tidal phenomena, nature of the beach, seasonal variation, animals and human presence [35]. The nature of the beach has an important effect on the pathological species which could exist there. Wet soil which has organic substances can be a favourable environment for enteric bacteria [26]. Mendes et al. [21] studied the effects of tides on the number of pathogens and faecal index microorganisms in the soil without finding any clear differences. However, Obiri-Danso & Jones [25] showed a very small difference for pathogens and faecal index microorganisms count after and before tidal phenomena. Moreover, in the same study campylobacters showed different counts with different seasons, with the highest rate in winter 100%, followed by spring where it reached around 33-67%. However, in autumn it increased again to around 67-78%. The lowest counts were found in summer.
Furthermore, when organisms move from one environment to another many factors including temperature, water availability, pH, and nutrient change. For instance, these microorganisms might be exposed to higher temperatures and salinity or receive little rainfall in their new environment. Some species can adapt to all the changes in the new environment while others cannot and thus would not survive. Some adaptations could cause microorganisms to be more potentially pathogenic or less productive than others in their environment.
In Abu Dhabi, the Corniche beach which is a new man-made beach had been recently developed in the northern coast of the city. Although construction developments had been designed based on an environmental framework which ensures sustainability [1] studies which examine the microbial profile of soil from this newly developed beach are still lacking. This study explored the difference between Abu Dhabi man-made and natural beaches in terms of soil microbial profile. Differences were examined with regards to soil water content and depth. To our knowledge, this is the first study which attempts to provide an idea about the presence of both potentially pathogenic and non-pathogenic microorganisms inhabiting the soil beach within Abu Dhabi.
2. Materials and Methods
2.1. Site Description
Sampling sites for collection of soil samples in this study were chosen to be man-made and natural. The man-made beach selected was the Abu Dhabi Corniche beach because it is not a natural beach. It is located along the north-western shore of the capital of the UAE. It is a newly developed beach that was awarded in 2009 to National Marine Dredging Company (NMDC) for recreational development. It consists of the development of a 3.5 km beachfront half of which is new [23]. It is protected from erosion caused by tidal forces with a 3.5 km submerged rock sill below the water, with sand in the top layer of beach equivalent to any internationally acclaimed beach. Beach sand was imported to that beach and about 3.5 million m3 were dredged from the sea bottom and used [23]. At different period of time, the sand had to be replaced and replenished with sand from other sources. Thus, many differences between natural and exotic microorganisms could have been introduced due to this process on the other hand, Al Yas beach which is located outside Abu Dhabi Island was used as natural resource of beach soil. This area does not undergo developmental changes and maintenance. Therefore, microorganisms would be assumed to live in their natural habitats. Both environments are characterized by hot weather, moisture and lack of rainfall during the summer. During winter, the weather becomes cooler and drier with a small chance of rainfall.
2.2. Sample collection and field measurements
Beaches were visited 2 times at 1-2 weeks intervals from November to December 2012 (Wheeler Alm., et. al., 2003). Sampling occurred over a 3 hour period between 12:00 pm and 3:00 pm (UAE local time) to control the differences in solar insulation [32]. The number of humans within 100 m during the sampling period was recorded. Also, air and soil temperature; and humidity were measured to set the ambient temperature [15]. General observations of the site were recorded, and pictures were taken for documentation. Sieves, gloves and sample containers were used to collect the soil samples from the beach. Wet (collected from the swash zone) and dry (collected away from swash zone) soil samples were collected from each site and the distance between the transects was recorded [2, 9]. This was done in order to compare the microbial profile of wet and dry soil samples in the same beach. Soil was collected from different depths (5 and 15 cm below the surface) by using manual soil-coring devices in order to preserve the integrity of the inhabiting microorganisms. This allowed collection of soil without mixing and hence measurement of each depth separately. Three sub-samples were collected for each sampling criteria to represent the beach as a whole. Those sub-samples were taken to ensure proper representation of the microbial profile of the whole beach. All collected and transported samples were placed in sterile; dry and cold environment (around 4 °C) to avoid contamination and further growth of inhabiting microorganisms. In addition, deep freezing was done to stabilize the microbial DNA used later for PCR.
2.3. Sample Analysis Strategies
2.3.1. Cultivation Analysis
General procedures of detecting microorganisms were followed using cultivation methods. A total of 16 samples were collected from both beaches depending on different parameters like water content and depth of soil (cm). Water content depended on distance from sea water and varied from 3-10m for wet soil and 20-24m for dry soil. The depth of the soil sample was fixed for 5 cm and 15 cm. Control samples of enterococci, yeast, faecal coliforms, Escherichia coli, Staphylococcus aureus, were inoculated in all types of growth media used. Enterococci, Escherichia coli, and Staphylococcus aureus were cultivated in MacConkey, EMB, Mannitol Salt Agar (MSA) and then incubated for 18-72 h at 30-35 °C [11]. Yeast and fungi control samples for Malt Salt and Sabouraud Dextrose Agar (SDA) were retrieved from the University of ADELAIDE mycology online database. Negative controls were made for all growth media and left for few days to test for growth. All media were prepared as per the manufacturer's instruction and sterilized.
Cultivation of soil samples followed the previously established method by Sabino, et. al., [30] with some modification depending on experimental circumstances. Bacteriological analysis started with having 6 tubes each containing 1 g of the soil sample it was was diluted in 2 mL of Luria broth and, agitated for 6 min at 500 rpm. Two different concentrations (0.25 mL and 1 mL) were later taken using sterilized pipettes, and poured each in separate MacConkey growth media. This step was repeated for all other growth media such as EMB, MSA, Malt Extract and SDA. Sterilized spreaders were then used to spread the liquid in the growth plates which were incubated at 37 °C for 24-72 hour. All bacterial, yeast and fungal growth analysis was reported as colony forming units (CFU) per g of soil. All the physical characteristics of the colonies and growth media were checked and noted.
2.3.2. Electrophoresis-based Polymerase Chain Reaction
Polymerase Chain Reaction (PCR) was used for more specific identification of some microorganisms based on the detection of unique gene sequences [22]. Preparation of PCR sample solution from beach soil samples followed TAKARA manual [34] for the bacterial screening PCR kit protocol 1.B. by preparing a heat extracted sample directly from bacteria. A small amount of the bacteria from the colony was extracted and suspended in 100 µL volume of Chelex solution. Then the mixture was heated at 99 °C for 5 minutes, this was quickly followed by cooling down by placing on ice for 1 minute or more. The mixture was later centrifuged at 12000 rpm for 1 minute. The centrifuged supernatant was used as the DNA sample solution for PCR.
PCR analysis of enterococci, Escherichia coli and Salmonella was performed according to the TAKARA manual [34]. The 2X premix solution, primer mix ENT, DNA sample solution were all mixed together for analysis. The Negative controls were also subjected to PCR using the specific primers and dH2O instead of DNA sample solution. Moreover, positive controls were amplified as per the instructions specified by the kit. ENT primers were used to identify Enterobacteriaceae family including E. coli. PCR primers were used for targeting specific Enterobacteriaceae 16S rRNA gene markers (Figure1). Thermal cycler (PXE 0.2 Thermal Cycler) was used for amplification, and the PCR parameters were as such: 95°C for 60 seconds, then 95°C,59°C, 72°C all for 30 seconds, finally 72°C, 4°C respectively for 60 seconds.
Following PCR, gel electrophoresis was used to identify the bands specific for each species family or genus. Agarose and TBE buffer preparation was according to the TAKARA manual [34] for bacterial screening PCR kit. In this experiment, 15 µl of PCR amplicons were added to 3 µL of loading buffer. Promega 100bp DNA Ladder was used as a marker to indicate the size of DNA bands. A 1.8% agarose gel was run at 65 V for 40 minutes and then stained in ethidium bromide and finally placed under UV transilluminator. A band of approximately 420 bp evident with the ENT primers indicated that the samples contained a member of the Enterobacteriaceae family including E. Coli.
3. Results
3. 1. Cultivation Analysis
Yas beach is a natural beach located outside Abu Dhabi Island and is considered as the control beach for this study. The average ambient temperature of the two trials was 31°C and average humidity was 60%. Results from the 2 trials for Yas beach showed a mean growth) value of 11.71± 13.81CFU/g of soil. Moreover, soil samples from this beach showed growth in Mannitol Salt Agar (MSA) which was indicative of the presence of Staphylococcus aureus and other Staphylococci. Growth on MacConkey medium indicated the possible presence of Pseudomonas aeruginosa, Shigella, Enterobacter and Klebsiella in Yas beach soil. Fungal and yeast cultivation results revealed the likely growth of Candida guilliermondii, Candida parapsilosis, Trichophyton equinum, Trichophyton terrestre, Malbranchea sp., Aspergillus fumigates, Trichophyton schoenleinii Acremonium sp. and Apophysomyces elegans (Table 1).
Table 1. Average Microbial Growth (CFU/g) for Yas Beach Soil Samples (N=2)
| Criteria | Mannitol Salt | MacConkey | EMB | Sabouraud Dextrose | Malt Salt | |
| Depth | Water content | |||||
| 5 cm | ||||||
| Dry | 4 | 31 | 2 | 1 | 3 | |
| Wet | 7 | - | - | - | 8 | |
| 15 cm | ||||||
| Dry | 2 | 2 | - | 1 | - | |
| Wet | 5 | 33 | - | 38 | 27 | |
Dhabi Corniche beach which is a man-made beach is located on the north-western shore of Abu Dhabi island. The average ambient temperature of the two trials was 27 °C and average humidity was 60%. Results from the examination of 8 samples of soil (both wet and dry) from the Corniche beach showed a mean growth value of 21.57 ± 27.73 CFU/g of soil. It revealed growth with a total of 47.5%. MacConkey and MSA plates revealed significant growth (CFU/g) especially for wet soil. The minimum growth average value was 7 CFU/g of soil while the maximum value was 38 CFU/g of soil. Staphylococcus aureus, Pseudomonas aeruginosa, Shigella, Enterobacter, Salmonella and E. coli all potentially existed in Corniche beach soil. For yeast and fungi, the possibilities were Malbranchea sp., Aspergillus niger, Trichophyton equinum, Paecilomyces variotii, Aspergillus carbonarius and Candida glabrata (Table 2).
Table 2. Average Microbial Growth (CFU/g) for Corniche Beach Soil Samples (N= 2)
| Criteria | Mannitol Salt | MacConkey | EMB | Sabouraud Dextrose | Malt Salt | |
| Depth | Water content | |||||
| 5 cm | ||||||
| Dry | 13 | 5 | - | 15 | 4 | |
| Wet | 40 | 88 | 3 | 25 | 2 | |
| 15 cm | ||||||
| Dry | 3 | 64 | - | - | 1 | |
| Wet | 70 | - | - | - | 38 | |
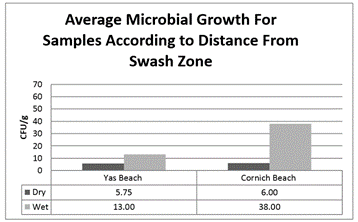
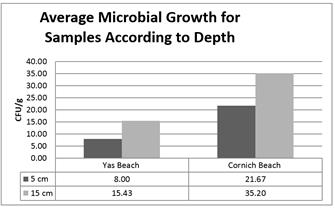
The average growth (CFU/g) for wet and dry soil isolates from Corniche beach (38 ± 32.19 CFU/g and 6.00 ± 5.69 CFU/g, respectively) was 10-60% higher than that for isolates from Yas beach (13.00 ± 14.69 CFU/g and 5.75± 10.25 CFU/g, respectively, figure 3). Moreover, the average growth (CFU/g) for isolates for 5 and 15 cm depths from Corniche beach (21.6 ± 27.82 CFU/g and 35.2 ± 32.61 CFU/g, respectively) was 50-70% higher than that for isolates from Yas beach (8 ± 10.46 CFU/g and 15.43 ± 16.48 CFU/g, respectively, figure 4).
3. 2. Electrophoresis-based Polymerase Chain Reaction (PCR)
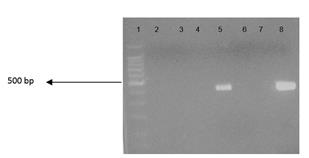
The PCR analysis was performed to confirm the presence of Enterobacteriaceae family strains in soil samples from either beach. Samples used for PCR and electrophoresis were all gram negative bacteria from MacConkey and EMB growth media. Figure 2 shows the matching in size for the positive control in lane 8 and samples from MacConkey plate in lane 5. The results showed positive growth for bacteria isolated from MacConkey plate from Corniche beach soil samples indicating the presence of E. coli, salmonella and coliforms in the soil. However, the other sample from MacConkey from Yas beach showed a negative result for coliforms using the same ENT primers which means that other gram negative bacteria present. Moreover, in another electrophoresis gel, wet sample from the Yas beach (MacConkey, 15 cm depth, 1 ml concentration) demonstrated also negative result for coliforms using the same ENT primers. This also suggested that other gram negative bacteria (but not E. coli) could be present.
4. Discussion
Results of the study showed some similarities between bacteria that inhabited both man-made and natural beaches. The most frequent gram positive isolates were species of Staphylococcus, whereas gram-negative genera from both beaches were Pseudomonas, Enterobacterriaeae sp. The only difference between the two beaches was with regards to E. coli which was detected in all samples from Corniche beach but only in one sample from Yas beach. E. coli could have inhabited the soil from human skin, infection or animal. In a study done by Anderson et al., [3], similar findings were reported about E.coli and the reason was suggested to be the increase of beach users. In Corrniche beach there was at least 1 to 2 humans each 5m3 whereas in Yas beach there was none. Moreover, growth of the algae Cladophora could have provided a favourable environment for the stability and growth of E. coli [10]. This could be a reason for the greater growth of E. coli in Corniche beach. An additional cause could be related to the source of the soil itself. E. coli could be considered a health risk to beach users. Moreover, Staphylococcus sp. outnumbered other bacteria in all soil samples. Dowidart & Abdel-Monem [8] found that Staphylococcus sp. predominate over other flora in the sand. Classification of the study indicated a total of 20-30% growth for S. aureus, 70-80% growth for other Staphylococcus sp. Similarly, a study done by Prado et al., [27] on two popular beaches in Chile revealed 24% growth as S. aureus and 76% as other Staphylococcus sp. The presence of these bacteria could be related to human activity. Corniche beach showed more CFUs for Staphylococcus sp. as it had more frequent human activity as compared to Yas beach. Papadakis et al., [26] demonstrated that the presence of S. aureus correlated with the presence of yeasts of human origin in soil samples. This was related to this study findings as yeast was isolated from samples taken from both Corniche and Yas beaches.
Regarding results of yeast and fungal growth, yeast like Candida sp. were found in 1.25% of samples from both beaches, whereas Candida albicans was not detected at all. Other researchers also had reported the absence or low incidence of C. albicans [28, 12, 4]. Furthermore, in this study all yeast from both beaches were Candida species which is comparable to results found in a research study done by Sabino, et. al. [30] which demonstrated that 67.5% of the detected yeast were Candida species. Other fungi were C. guilliermondii and C. parapsilosis; which were also isolated by other researchers from seashore soil [4]. Moreover, comparison of the fungal profile growth in media plates between man-made (4%) and natural beach (6%) indicated that the man-made beach had less growth than natural beach. This could be attributed to the low level of human activity and soil maintenance in Yas beach (natural) in contrast to Corniche beach (man-made) which is subjected to constant maintenance and has laws to prohibit the presence of animals. Related to the safe CFU/g threshold, based on Sabino, et. al. [30] study, 15 CFU/g, is the calculated threshold for the yeasts. In this study, all the isolated yeast showed less than 15 CFU/g, therefore, would not be considered as a health risk for beach users.
Potentially pathogenic fungi such as Aspergillus sp. were found in more abundance in wet soil samples as compared to dry soil samples probably because fungi have higher resistance to solar exposure compared to bacteria [3]. A study by Izquierdo et al., [16] revealed that saprophytic fungi such as A. Fumigates, A. Candidus and A. ochraceus were isolated from wet soil areas of the beach in high tidal conditions. Whereas in other investigations such as that of Sabino, et.al., [30] these fungi, were found in dry soil with high average growth of CFU/g than what was found in our study for Yas and Corniche soil beaches. Furthermore, Trichophyton sp. showed higher growth percentage than yeast and bacteria and was detected in all the samples collected. Salvo, & Fabiano, 2007 in Italy, Ulfig, et al., 1997 in Spain, Vidal, et al., 1966 in Sweden and Sabino et. al. in Portugal all showed the same conclusions for this species. Also, Harris et al. 1995; Bater 1996; Van der Heijden et al. 1998; Caruso et al. 2008 indicated that the growth of fungi could be caused by the extinction of the native species [14]. Hence, this could be a reason for the disappearance of some significant species from beach soil such as E. coli in Yas beach. It has been said by Sabino, et. al. in 2011 that 17 CFU/g is the threshold for potentially pathogenic fungi, and 8 CFU/g for dermatophytes. In comparison, both beaches (man-made and natural) had higher number of CFU/g of soil hence suggesting a potential health risk for human health. More studies are needed however to confirm or generalize this finding.
The hypothesis of the study suggested that the growth of microorganisms in man-made beach soil would be higher as compared to natural beach soil. Results of the study showed a high average growth CFU/g for Corniche beach for both parameters (water content and depth from the soil surface). The results demonstrated that average growth was higher in wet soil as compared to dry soil. This suggested the existence of a correlation between water content and growth of bacteria in soil. Our study is supported by two other studies which showed the same results. Su, Jianping. et. al., 2004, found that wetlands showed more abundant microbial population as compared to dry lands. Also, Kaneko and Atlas, [17] found that the density of bacterial communities positively correlated with the water content in the soil. Therefore, the studied man-made beach has both similarities and differences compared to natural beaches with some differences the natural beach of the study. Many reasons support this finding as one of the microbial growth features is the availability of water content [24]. Papadakis et al., [26] stated that the wet soil is enriched with organic substances which provide a harbor environment for enteric bacteria allowing it to survive longer than in other areas. The natural beach of the study might have been affected by the surrounding construction which might have in turn affected the microbial population living there. However, a study performed by Brandão et al., [5] showed that the microbiological growth was higher in dry soil as compared to wet soil. This could be due to the preferences of beach users who often enjoy sitting in the dry soil more than the wet soil.
The other parameter used in this investigation between the man-made beach and the natural beach was the depth of the soil from the surface. The finding of this study showed a higher average growth of microorganisms in the depth of around 15 cm. That finding is supported by a study by Papadakis et al., [26] which stated that enteric bacteria survive for short duration on the surface of dry soil as it gets destroyed mostly by environmental pressure. Therefore, its favorable conditions are in deeper layers of soil. Also, it was stated by Othman, 2003 that variations in temperature of the soil occur in surfaces and deeper depths. Soil in deep depth does not exhibit very dramatic daily and seasonal variations in temperature, so growth of microorganisms, gets more abundant and stable [13]. However, the only exception was that E. coli was isolated only from the upper 5 cm of soil in this study, while growth in the depth of 15 cm was not detected. This could be related to the need of this particular species to sunlight and oxygen which are found abundantly in the upper layers of soil. This finding is supported by the Kleinheinz, [19] and Desmarais, et. al., [7] who demonstrated that with deeper depth, microorganisms become less abundant.
PCR results demonstrated the presence of E. coli, Salmonella and coliforms in Corniche beach but not in Yas beach for the same species. The presence of members of the Enterobacteriaeae species could indicate recent faecal pollution [14] Therefore, this could be an indication of some pollution in the Corniche beach. A study by Hafez, E, & Elbestawy, E., [14] demonstrated that the dominant gram negative bacteria in the soil of a number of beaches they assessed in their study were identified as Shigella flexneri and Escherichia sp. using 16S rRNA primers. However, direct extraction from soil was used in the previous study while in this study manual DNA extraction from culture was utilized. PCR was used in the identification of human enteric pathogens in two South-western Lake Michigan recreational beaches. The results indicated the presence of Salmonella (0.7% of isolates) and Campylobacter (22.7%) [18]. Our results similarly revealed that Salmonella, which is potentially pathogenic to human health, inhabited Corniche beach soil.
5. Conclusion
In conclusion, the man-made beach (Corniche beach) showed a higher microbial growth in terms of water content and depth from the soil surface as compared to the natural beach (Yas beach). Furthermore, E. coli and coliforms had been detected in Corniche beach only. So regardless of the great effort invested for maintaining a good soil quality in man-made beach, there could be some factors which allow microbes to grow and thrive in the soil of such beaches. Even if these microorganisms have been introduced unnaturally to the beach by different means including the collection of soil from various locations and contamination from the skin/infection of humans and animals, they could adapt to the new environment with time and become part of the ecological soil diversity. Future research studies could investigate whether there could be differences within the soil microbial diversity over a longer period of time taking into consideration the seasonal and environmental changes.
Acknowledgement
Especial thanks to the NSPH Department at Zayed University for the help and support throughout the study.
References
[1]Abu Dhabi Urban Planning Council (AD-UPC). (2007). Plan Abu Dhabi 2030 [PDF]. View Site
[2] Alm, E., Burke, J., & Spain, A. (2003). Fecal indicator bacteria are abundant in wet sand at freshwater beaches. Water Research 37, 3978-3982. View Article
[3] Anderson, J.G., Rowan, N.J., MacGregor, S.J., Fouracre, R.A., Farish, O., 2000. Inactivation of food-borne Entero pathogenic bacteria and spoilage fungi using pulsed-light. IEEE Transactions on Plasma Science 28, 83-88.View Article
[4] Boiron, P., Agis, F., & Nguyen, V.H. (1983) Study of yeast flora of medical interest on the beach of Saint Anne in Guadeloupe. Bulletin de la Societe de Pathologie Exotique and its Subsidiaries, 76(4): 351-356. View Article
[5] Brandão, J.C., Veríssimo, C., Rosado, M.L., Sabino, R., Falcão, M.L., Giraldes, A., Rosado, C., Simões, M., Noronha, M.G., 2002. Final report of the project'' Microbiological Quality of Coastal Beaches sands,'' European Blue Flag Association View Article
[6] Burton, J.R., Gunnison, D., Lanza, G.R. (1987). Survival of potentially pathogenic bacteria in various freshwater sediments. Applied Environmental Microbiology 53, 633-638. View Article
[7] Desmarais, T.R., Solo-Gabriele, H.M., &Palmer, C.J. (2002). Influence of soil on fecal indicator organisms in a tidally influenced subtropical environment. Appl Environ Microbiol, 63(3):1165-72. View Article
[8] Dowidart, A., Abdel-Monem, M.H. (1990) Effect of chemical pollutants on bacterial counts in El-Temsah Lake area, Ismailia, Egypt. Journal of the Egyptian Public Health Association,65(3-4), 305-328. View Article
[9] Elmanama, A. A., Fahd, M. I., Afifi, S., Abdallah, S., & Bahr, S. (2005). Microbiological beach sand quality in gaza strip in comparison to seawater quality. Environmental Research, 99(1), 1-10. View Article
[10] Englebert, E. T.; McDermott, C.; Kleinheinz, G. T. (2008). Effects of the nuisance algae, Cladophora, on Escherichia coli at recreational beaches in Wisconsin. Sci. Total Environ., 404 (1), 10-17. View Article
[11] European Pharmacopeia. (n.d). Dehydrated culture media. 3, 217-220,577-579.
[12] Figueras, M.J., Guarro, J., Soler, L., Inza, I., Polo, F., 1992. A pilot study on the contamination of Catalounian beaches. National Environmental Congress, Madrid.
[13] Gibbali,M.A. (1988) Studies on the flora of northern Sinai. M.Sc. Thesis. Fac. Agric., Cairo, Univ.,Egypt. 393.
[14] Hafez, E, & Elbestawy, E. (2009). Molecular characterization of soil microorganisms: effect of industrial pollution on distribution and biodiversity. World Journal of Microbiology and Biotechnology 25. 2, 215-224. View Article
[15] Hurst, Christon J. (Editor); Garland, Jay L. (Editor); Mills, Aaron L. (Editor). Manual of Environmental Microbiology (3rd Edition). Washington, DC, USA: ASM Press, 2007. p 8- 9,37.http://site.ebrary.com/lib/zu/Doc?id=10381779&ppg=31 View Book
[16] Izquierdo, J., Piera, G., Aledany, M.C., & Lucena, F. (1986) Estudio de la flora fungica de la arena de la playa de Barcelona. [A study of the fungal flora of the beaches in Barcelona.] Athens, United Nations Environment Programme, Mediterranean Action Plan, Mediterranean Marine Pollution Monitoring and Research Programme (MED POL Research Project Final Report).
[17] Kaneko, T., Atlas, R. (1977). Diversity of bacterial populations in the Beaufort Sea. Nature 270, 596-599. View Article
[18] Kinzelman, J.; McLellan, S. L.; Amick, A.; Preedit, J.; Scopel, C. O.; Olapade, O.; Gradus, S.; Singh, A.; Sedmak, G. (2008) Identification of human enteric pathogens in gull feces at Southwestern Lake Michigan bathing beaches. Can. J. Microbiol., 54 (12), 1006-1015. View Article
[19] Kleinheinz, G. T. (2008). Comprehensive beach sanitary survey project in Wisconsin. University of Wisconsin, USA View Article
[20] Mancini, L., D'Angelo, A.M., Pierdominici, E., Ferrari, C., Anselmo, A., Venturi, L., Fazzo, L.,
[21] Mendes, B., Nascimento, M.J., Oliveira, J.S., 1993. Preliminary characterisation and proposal of microbiological quality standard for soil beaches. Water Science Technology 27(3-4), 453-456. View Article
[22] National Center for Biotechnology Information (NCBI). (n.d). PCR. Retrieved Nov 15, 2012, from View Article
[23] National Marine Dredging Company (NMDC). (n.d). Corniche beach, Abu Dhabi, UAE. Retrieved June 6, 2012 fromhttp://www.nmdc.com/index.php?cmd=cms__view&article_id=95 View Site
[24] Nester, E., Anderson, D., Roberts, C., & Nester, M. (2007). Microbiology: a human perspective. New York : McGrow-Hill. View Book
[25] Obiri-Danso, K., & Jones, K. (1997) Seasonality of thermophilic Campylobacter and faecal indicators in sediments from the intertidal zone of Morecambe Bay. In: Proceedings of the 9th International Workshop on Campylobacter, Helicobacter and Related Organisms. Cape Town, 15-19 September.
[26] Papadakis, J.A., Mavridou, A., Richardson, S.C., Lambiri, M., & Marcelou, U. (1997) Bather related microbial and yeast populations in soil and seawater. Water Research, 31(4), 799-804.
[27] Prado, B., Bernal, P., Contreras, M., Savedra, M., del Moral, A., & Joyas, M. (1994) Numerical taxonomy of staphylococci isolated from water and beach soil from Valparaiso and Vina del Mar, Chile. Revista Iberoamericana de Microbiologia, 36(2), 71-77. View Article
[28] Roses, C. M., Isern, A.M., Ferrer, M.D., &Fernandez, P. F. (1988) Microbiological contamination of the soil from the Barcelona city beaches. Revista de Sanidad e Higiene Publica, 62(5-8), 1537-1544. View Article
[29] Roach, J. (2005). Beach bacteria warning: that soil may be contaminated. Retrieved April 17, 2012 from View Article
[30] Sabino, R., Veríssimo, C., Cunha, M. A., Wergikoski, B., Ferreira, F. C., Rodrigues, R. (2011). Potentially pathogenic fungi: An unacknowledged risk at coastal resorts? new insights on microbiological soil quality in Portugal. Marine Pollution Bulletin, 62(7),15061511.doi:10.1016/j.marpolbul.2011.04.008 View Article
[31] Sato, M.I.Z., Di Bari, M., Lamparelli, C.C., Cristina, T.A., Coelho, M.C.L.S., Hachich, E.M. (2005). Sanitary quality of sands from marine recreational beaches of São Paulo, Brazil. Brazilian Journal of Microbiology, 36, 321-326. View Article
[32] Shah, A. H., Abdelzaher, A. M., Phillips, M., Hernandez, R., Solo‐Gabriele, H. M., & etc. (2011). Indicator microbes correlate with potential pathogenic bacteria, yeasts and helminthes in soil at a subtropical recreational beach site. Journal of Applied Microbiology, 110(6), 1571-1583. View Article
[33] Soussa, M.L.R. (1990) Micoses. [Fungi.] Lisbon, National Institute of Health (INSA), Centre of Epidemiologic Surveillance of Transmissible Diseases (Epidemiology Bulletin No. 5).
[34] TAKARA. (n.d). Bacteria screening PCR kit. From View Site
van Veen, J., van Overbeek, L., & van Elsas, J. (1997). Fate and activity of microorganisms introduced into soil. Microbiology and Molecular Biology Reviews, 61(2), 121-135. View Article
[35] World Health Organization (WHO). (2003). Guidelines for safe recreational water environments, 1, Coastal and Fresh Waters. World Health Organization, Geneva. View Article

